Endogenous Retroviruses in Fighting Against Radiation-Induced Inflammation
THE PROBLEM
Moderate and even low radiation doses may induce acute or chronic inflammatory response that disrupts organ functions and are an important factor of secondary radiation-related disorders, such as vascular abnormalities, neuronal damage, autoimmune diseases and radiation-related cancer. Molecular patterns associated with activation of viral genomes, such as viral RNAs and proteins are shown to be essential factors that exacerbate inflammatory response. The ongoing projects in the lab are focusing on the role of endogenous retroelements, particularly retroviruses, in molecular mechanisms of innate immune response, monocyte/macrophage differentiation and inflammation in reaction to radiation-induced cellular stress.
"The translational impact of our studies is in defining the role of endogenous retroviruses as a potential target for pharmacological intervention and programmable gene modulators against radiation-induced inflammation."
WHAT WE'RE DOING
Currently we are focusing on two research topics related to this problem.
Role of Human Endogenous Retroviruses (HERVs) in Radiation-Induced Inflammation and Cell Pathogenesis
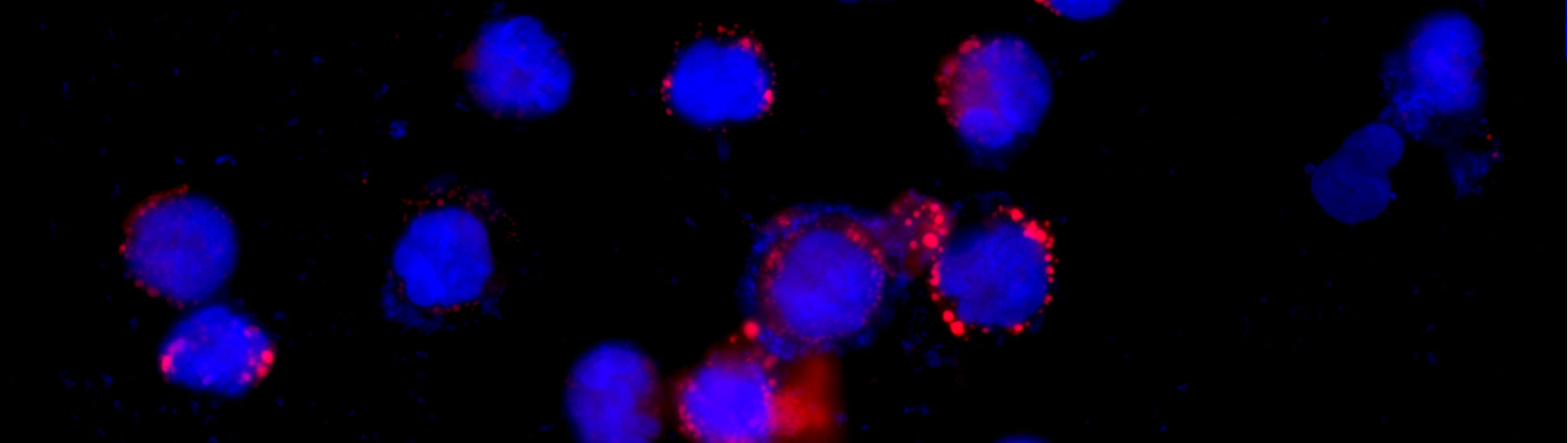
We analyze the effect of gamma radiation on the expression of HERVs (8.3% of our genome) and their impact on inflammatory response in various types of immune cells. We found that promoters of certain HERV genomes become permanently activated after the single doses of radiation. Retroviral RNA as well as particular viral proteins, especially Env (envelope protein), affect innate immune signaling and can enhance radiation-induced inflammation via activation of type I interferon pathway and production of proinflammatory cytokines.
HERVs in Intercellular Communications between Radiation-Exposed and Unexposed Cells – Role of Extracellular Vesicles
We investigate extracellular vesicles (EVs), particularly exosomes, released from radiation-exposed cells, and their involvement into the response to irradiation in unexposed tissues. As intercellular communicators, EVs transfer molecular signals, such as mRNA, microRNA, viral RNA and proteins from irradiated cells to the unexposed bystander cells, activate certain signaling pathways and change gene expression profiles in the recipient cells. Therapeutic modulation of exosome release can reduce indirect pathogenic effect of radiation exposure to the unexposed tissues and organs.
LOOKING AHEAD
Ultimately, we hope to elucidate retrovirus-related mechanisms that enhance radiation-induced inflammation and affect pathogenic effect of radiation on unexposed cells. Our long term goal is to manage the inflammatory response to ionizing radiation using reprogramming cytokine profile of radiation-activated cells via the modulation of expression of endogenous retroelements. The translational impact of our studies is in defining the role of endogenous retroviruses as a potential target for pharmacological intervention and programmable gene modulators against radiation-induced inflammation.