THE PROBLEM
Traumatic brain injury (TBI) and post-traumatic stress disorder (PTSD) are among the most common conditions affecting veterans and military populations. Learning the molecular pathway underlying neural progenitor - vascular interaction permits the development of new therapeutic interventions for TBI. Information acquired on molecular controls for genome instability in cortical neurons may shed light on prevention of PTSD.
Research projects in Dr. Feng’s lab originated from studying human genetic disorders that impair cerebral cortical development. These disorders are caused by abnormal generation of neurons from neural progenitor/stem cells and have diverse clinical manifestations, ranging from profound mental retardation to epilepsy and mental illnesses. By studying genetic variances associated with defects in neural progenitor function, the study will improve the understanding of the genetic and developmental basis of neuropsychiatric disorders.
- Current funding support: NINDS/NIH, USU
- Previous funding support: NIMH/NIH, NICHD/NIH, Brain Research Foundations
"We study mechanisms by which neurons are generated in the cerebral cortex, the organ responsible for our intellectual activities. Our work sheds light on the molecular genetic basis of neurological and psychiatric disorders."
OUR APPROACH
By combining innovative cell and mouse models with high throughput genomics and proteomics approaches, the lab has investigated the mechanisms of these cortical developmental disorders and identified new pathways underlying the regulation of neural progenitor/stem cells-vascular interaction and neuronal genome stability protection. These studies may provide novel insight into the pathogenesis of a wide variety of neurological and mental disorders as well as neurodegenerative diseases.
RESEARCH DISCOVERIES
Our results have provided novel insight into the etiology of neuropsychiatric disorders including PTSD. They also shed light on the treatment of TBI through induced neural progenitor stemness via manipulating neurovascular interactions.
tNeural progenitors in the developing central nervous system make individualized fate decisions based on their spatial location with respect to the midline signal centers.
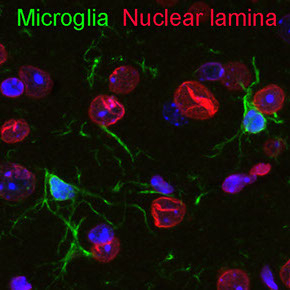
Bi-directional communication between neural progenitors and brain vasculature determines neurogenic plasticity.
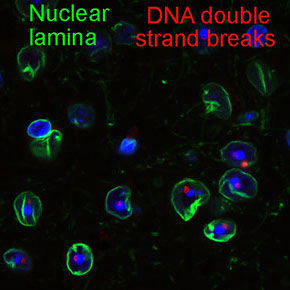
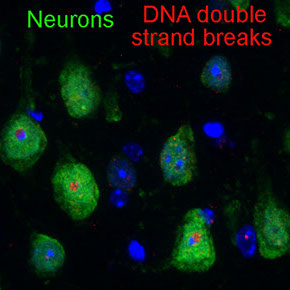
Proper replicating and remodeling heterochromatin in neuronal differentiation is essential for maintaining genome stability of cerebral cortical neurons.